For Patients
Ovarian tumor
Symptoms
Ovarian tumors are generally asymptomatic when they are small and rarely interfere with daily activities. Most women with ovarian tumors have regular menstruation, and the tumors do not significantly affect pregnancy. It is not uncommon that ovarian tumors are found by chance during uterine cancer screening or visiting internal medicine. Some women visit doctor and are diagnosed with the cancer after feeling tighter fit of their skirts or pants in the waist, while many others do not care the feeling by believing it due to being fatter.
Ovarian tumors can become as large as 20 cm or more in diameter. When the tumor grows, it can compress the bladder and the rectum, causing frequent urination and constipation. It can also cause the edema in the lower limbs due to compressed lymphatic vessels and impaired venous return (poor return of venous blood from the end of the lower limbs to the heart). Retention of ascites further increases abdominal circumference, which may cause forward protruding belly like a pregnant woman. When the base of the ovarian tumor is twisted (torsion of ovarian tumor pedicle) or part of the ovarian tumor ruptures, severe lower abdominal pain may occur, requiring emergency surgery.
Diagnosis
Since the ovaries are located in the abdominal cavity, the size and shape of the ovaries as well as the presence of adhesions are examined in pelvic examinations. The sizes and internal conditions of the ovaries are observed by transvaginal ultrasonography. In addition, imaging tests such as CT and MRI are concomitantly used to estimate their relationship with other organs such as the uterus, bladder, and rectum, the conditions inside the tumor, and the presence of swollen lymph nodes to estimate whether the tumors are benign, borderline malignant, or malignant. Definitive diagnosis (benign, borderline malignant, or malignant) is achieved through histological diagnosis (microscopic confirmation) of the samples obtained from surgery. Usually, approximately 90% of ovarian tumors are benign while approximately 10% of them are malignant. Distinguishing between benign and malignant tumor can be difficult by diagnostic imaging alone. In these cases, rapid tissue diagnosis may be used during surgeries.
When treating ovarian tumors (borderline malignant or malignant), it is extremely important to examine the spread of disease in order to decide on courses of treatment and hopes for treatment (prognosis). Since accurate determination of the detailed spread of disease is difficult only by imaging tests such as abdominal palpation, pelvic examination, ultrasonography, CT, and MRI, surgery is performed to achieve detailed intraperitoneal search and to check the presence of retroperitoneal lymph node metastasis.
The spread of disease is roughly classified into four stages from I to IV. Stage I represents "the state where the cancer is limited to the ovaries", stage II "the state where the cancer has spread into the pelvic cavity, namely, the spread to the uterus, oviduct, rectum, and the surface of the bladder (peritoneum)", stage III "the state where the cancer has spread beyond the pelvic cavity to the peritoneum in the upper abdomen, the omentum or small intestine, or alternatively metastasized to the retroperitoneal lymph nodes," and stage IV "the state where the cancer has metastasized to organs such as the liver and lungs (distant metastasis)." In 2014, the International Federation of Gynecology and Obstetrics (FIGO) published the "Postoperative stage classification (2014)." In Japan, a new postoperative stage classification (Acta Obstetrica et Gynaecologica Japonica (2014) FIGO 2014), which is in line with the international standards, has begun to be applied (Table 1).
Table 1 Postoperative stage classification of ovarian cancer (Acta Obstetrica et Gynaecologica Japonica (2014), FIGO2014)
| Stage I: Ovarian or intratubular development | ||||
|---|---|---|---|---|
| Stage IA | Tumors are confined to one ovary (no capsule rupture※1) or the oviduct, with no invasion to the capsule surface observed. No malignant cells are found in the cytodiagnosis of ascites or lavage fluid.※2 | |||
| Stage IB | Tumors are confined to both ovaries (no capsule rupture) or the oviduct, with no invasion to the capsule surface observed. No malignant cells are found in the cytodiagnosis of ascites or lavage fluid. | |||
| Stage IC | Tumors are confined to one or both ovaries or the oviduct, with either of followings observed: | |||
| Stage IC1 | Capsule rupture due to surgical operation | |||
| Stage IC2 | Spontaneous capsule rupture or invasion into the capsule surface | |||
| Stage IC3 | Malignant cells are found in ascites or during peritoneal lavage cytodiagnosis | |||
| Stage II: Tumors present in one or both ovaries or the oviduct, extending into the pelvis (small pelvic cavity), or primary peritoneal cancer | ||||
| Stage IIA | Progression and/or metastasis of tumors has extended to the uterus, the oviduct and/or the ovary | |||
| Stage IIB | Tumors have extended to other pelvic abdominal organs | |||
| Stage III: Tumor present in one or both ovaries or the oviduct, or primary peritoneal cancer with cytologically or histologically confirmed extrapelvic peritoneal dissemination and/or retroperitoneal lymph node metastasis | ||||
| Stage IIIA1 | Only positive for retroperitoneal lymph node metastasis (cytologically or histologically confirmed) | |||
| Stage IIIA1 (i) | Metastatic lesion with maximum diameters of 10 mm or less | |||
| Stage IIIA1(ii) | Metastatic lesion with maximum diameters exceeding 10 mm | |||
| Stage IIIA2 | Microscopic extrapelvic dissemination found with or without retroperitoneal lymph node metastasis | |||
| Stage IIIB | Intraperitoneal dissemination with maximum diameters of 2 cm or less found with or without retroperitoneal lymph node metastasis | |||
| Stage IIIC | Intraperitoneal dissemination with maximum diameters exceeding 2 cm found with or without retroperitoneal lymph node metastasis (including the extension to the liver and spleen capsule without parenchymal metastasis) | |||
| Stage IVDistant metastasis excluding peritoneal dissemination | ||||
| Stage IVA | Malignant cells found in pleural effusion | |||
| Stage IVB | Tumors with parenchymal metastases and the metastases to extraperitoneal organs (including the inguinal and extraperitoneal lymph nodes) | |||
Substances that increase in the blood due to the presence of tumors are determined and used to assist diagnosis. Such substances are commonly called tumor markers. At present, no marker has been found that specifically increases only in malignant tumors; therefore, several tumor markers are combined to be used to assist in diagnosis including recurrence as well as the assessment of therapeutic effects based on the knowledge of the characteristics of respective markers.
Treatments
- Benign tumor
Surgery is considered when the sizes of tumors exceed a predetermined level, or when solid tumors are found even if they are small. This does not apply if patients have symptoms such as lower abdominal pain. Emergency surgery may be necessary in case of sudden lower abdominal pain (due to ovarian tumor pedicle torsion and ovarian tumor rupture, etc.).
The choice of surgery is adnexectomy or cystectomy where only tumors are removed while preserving normal ovary for those who wish to become pregnant. Pregnancy is possible even if one ovary is removed. Laparoscopic surgery is also an option if preoperative examinations (including imaging tests such as ultrasonography, CT, MRI) do not reveal obvious evidence of borderline or malignant tumors. - Borderline malignant tumor
Most borderline malignant tumors show good treatment outcomes, but in rare cases, metastasis or recurrence may occur like malignant tumors, thereby requiring treatments similar to those for malignant tumors (ovarian cancer).
When lesions remain in the ovary (stage I), basic procedures such as bilateral adnexectomy, total hysterectomy, omentectomy, and intraperitoneal cytodiagnosis are considered. No postoperative chemotherapy is required when definitive diagnosis confirms that the tumor is of stage I. During intraperitoneal observations, peritoneal biopsies are performed for multiple sites to search for small lesions that can be confirmed by microscopic examination. For patients who wish to become pregnant, adnexectomy, omentectomy and intraperitoneal cytodiagnosis are conducted on the side where tumors developed unless preoperative imaging finds any obvious intraperitoneal abnormalities except for the ovaries, or unless there is obvious abnormal macroscopic finding during surgery. The probability of recurrence increases slightly unless basic procedures are used; therefore, employing basic procedures is considered once the patients no longer desire pregnancy.
When tumors are found to have extended outside the ovary (stage II-IV), tumor debulking are used depending on the spread of the disease in addition to basic procedures (bilateral adnexectomy, total hysterectomy, omentectomy and intraperitoneal cytodiagnosis). Systematic removal of retroperitoneal lymph nodes is not basically used unlike malignant cases; however, swollen lymph nodes with suspected metastasis may be removed for histological examination. When the spread of disease in the intraperitoneal peritoneum is suspected, the corresponding site is removed for histological examination (microscopic confirmation). When the spread of disease is found beyond the pelvic cavity or to the special peritoneum (invasive implant), chemotherapy similar to that for malignant tumor may be given.
As it is not clear yet whether treatments without basic surgical procedures (bilateral adnexectomy, total hysterectomy, omentectomy, and intraperitoneal cytodiagnosis) are acceptable for patients who wish to become pregnant and have lesions extending beyond the ovaries (stage II-IV), the patients should consult with their doctors carefully to determine the courses of treatment considering the risk of illness and the desire for pregnancy. - Malignant tumor (cancer)
The principle of the treatment for malignant tumors is to remove the tumors as much as possible by surgery and to eliminate residual tumors as much as possible (complete resection). After surgery, chemotherapy (anti-cancer drug treatment) is given in an attempt to completely eliminate residual tumors and tumor cells.
When the tumor is too large to remove appropriately in a single operation, an option is to remove only part of the tumor to be used for histological examination for definitive diagnosis and expect the effects of chemotherapy (anticancer drug treatment). Resection of secondary tumor is attempted after several rounds of chemotherapy.
Ovarian malignancies are considered to be among the solid tumors for which anticancer drugs are effective; thus, postoperative chemotherapy is required except in very early cases. In chemotherapy, two or three anticancer drugs, mainly paclitaxel, carboplatin, and cisplatin, are generally given periodically in combination. The drug to be used, administration method, dosage, and other factors are decided depending on patients’ conditions. Figure 1 shows 5-year survival rate of ovarian cancer patients in Japan by different stages.
Recently, the use of molecular-targeted therapies has become possible in combination with initial chemotherapy or as maintenance therapy after initial chemotherapy. Molecular-targeted therapies may also be used for chemotherapy for recurrent lesions. Please consult with your doctor for details. Chemotherapy (anti-cancer drug treatment) causes diverse adverse effects, such as decreased red blood cells, leukocytes, and platelets due to myelosuppression; impaired renal function, impaired liver function, hair loss, nausea, diarrhea, joint pain, and muscle pain. We attempt to address such symptoms with appropriate supplementary treatments. It is very important to describe the adverse effects at home after undergoing chemotherapies. Descriptions of the types and times of the adverse effects occurred as well as how long and how strongly they continued serve as valuable resources when consulting with doctors.
Figure 1. Five-year survival rate by the stages of ovarian malignancies for the cases diagnosed in 2012.
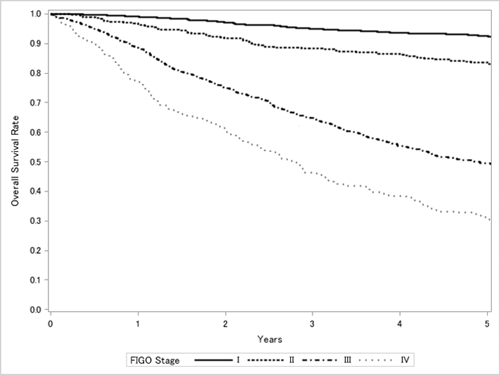
Report from the Gynecologic Oncology Committee of the Japan Society of Obstetrics and Gynecology, the 60th Annual Treatment Report
(Acta Obstetrica et Gynaecologica Japonica (2019) 71, 780)